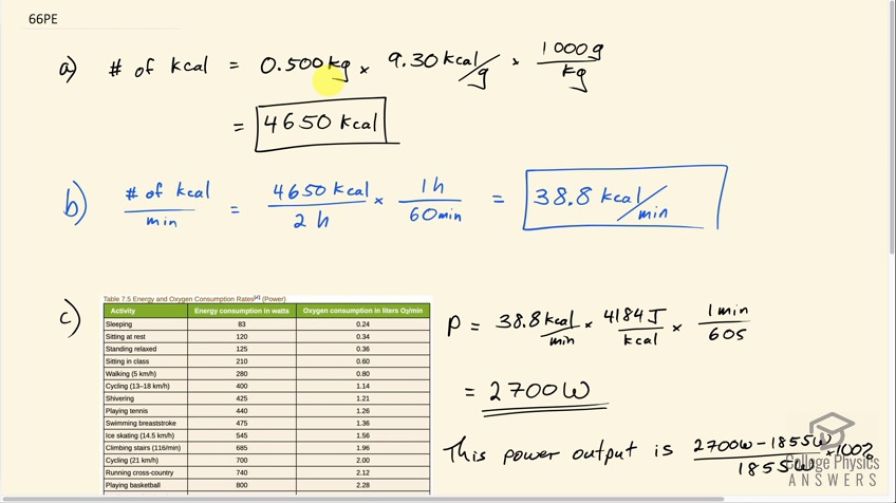
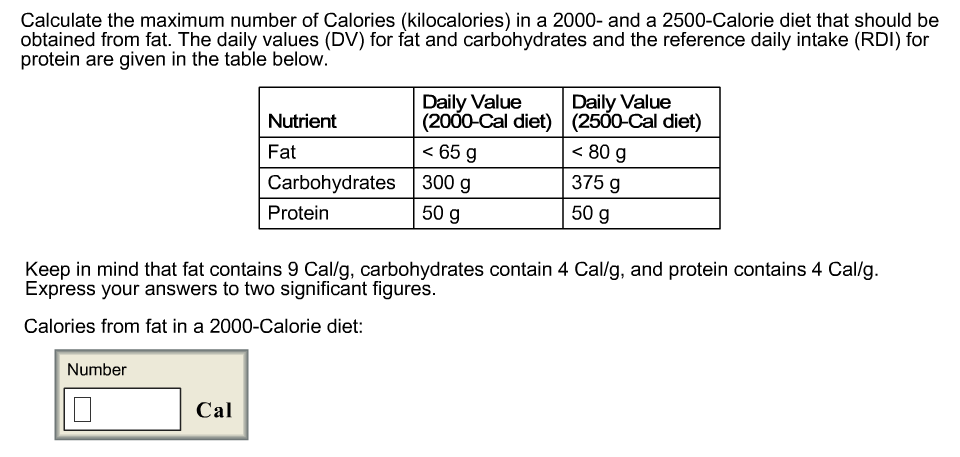
SOLVED: 22. Calculate the number of Calories of heat absorbed by the water (Q) for each of the food samples using the equation below and record in Data Table 2. Q =

SOLVED: Use the heat equation, 4-mCAT, to calculate the energy in kcal used to heat 3.4 kg of water from -26.9°C to 68°C. Note: (1 cal = 4.184 J, 1 calorie = 4.184 J)