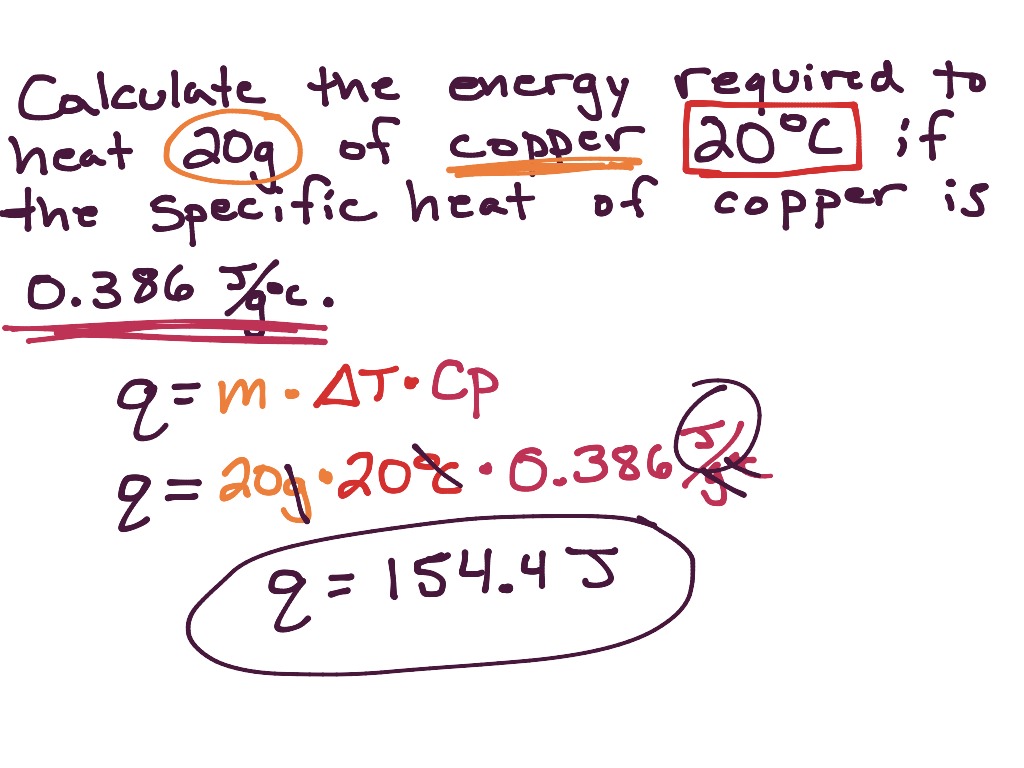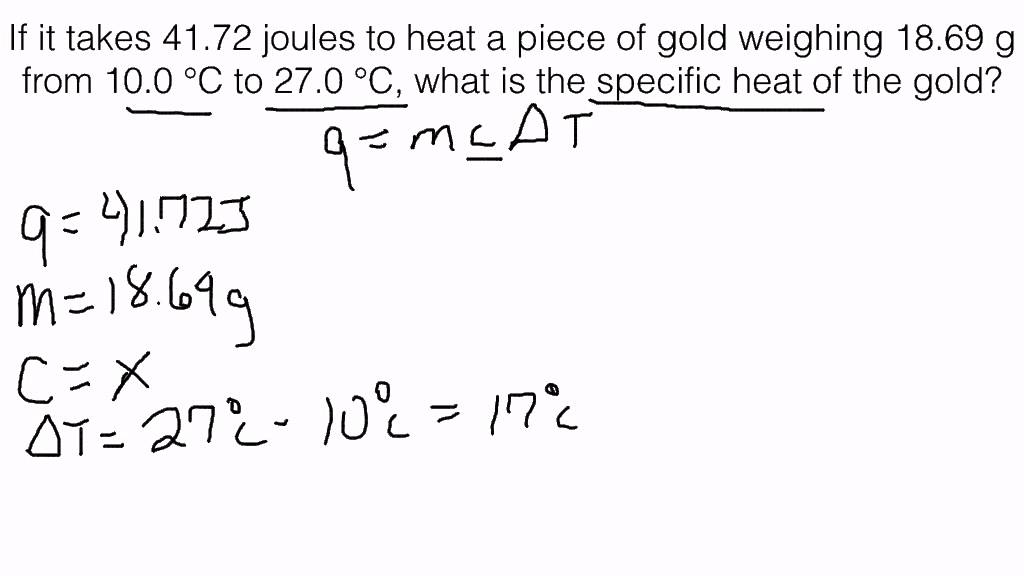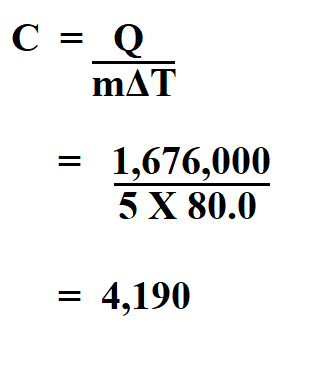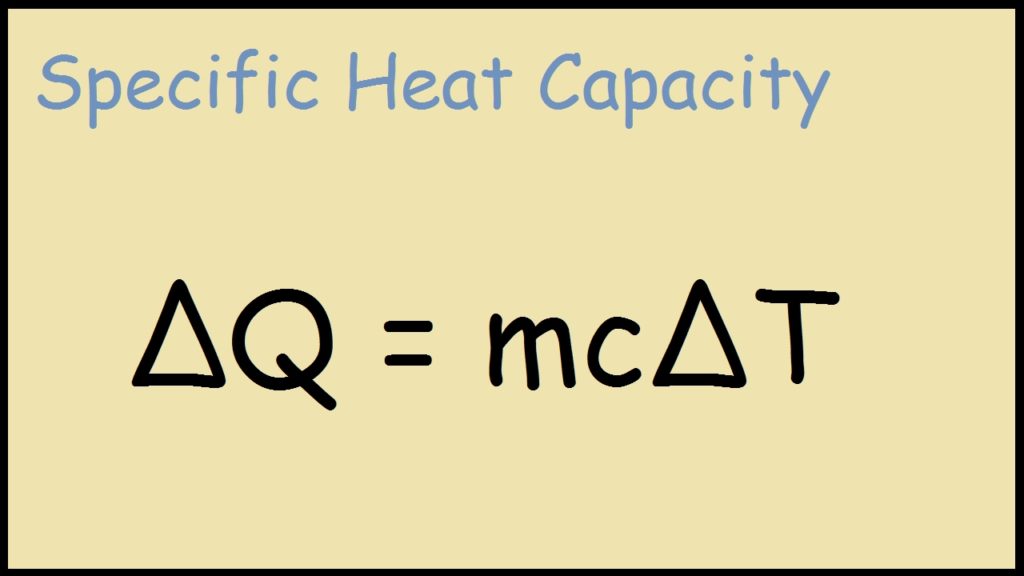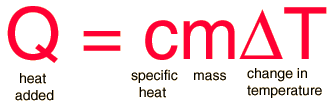45 g of water at 50∘C in a beaker is cooled when 50 g of copper at 18∘C is added to it.The contents are stirred till a final constant temperature is reached.Calculate

Heat Capacity and Specific Heat Calculations - Practice - A 1 g sample of decane, C 10 H 22 , is - Studocu
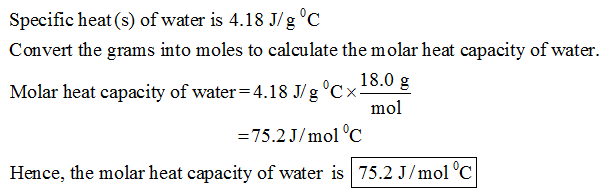
The specific heat of water is 4.18 J/(g⋅∘C). Calculate the molar heat capacity of water - Home Work Help - Learn CBSE Forum
Calculate the difference between between the principal specific heat capacity of 1g He at STP (R = 8.13 J/K Mol, J = 4.186 J /cal and molecular weight = 4 > | EduRev Class 11 Question
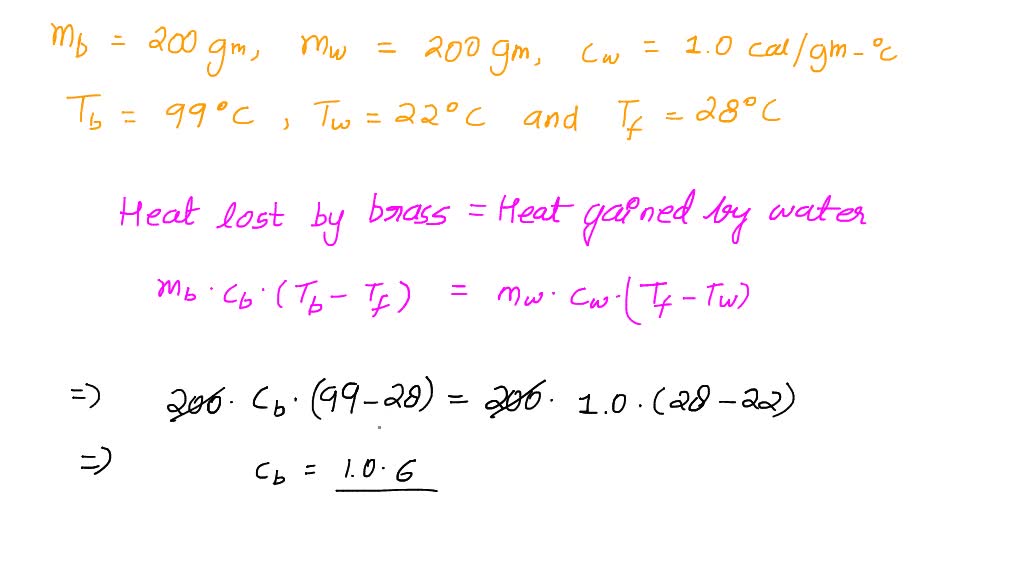
SOLVED: Calculate the specific heat of brass, given the following: T (hot) = 99 °C T (cold) = 22 °C T (final) = 28 °C (the brass lost heat and the water