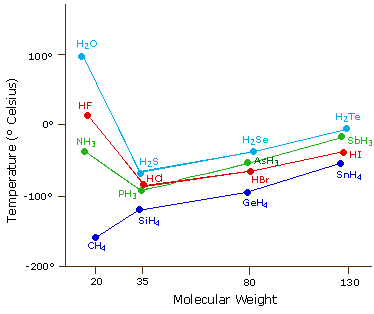
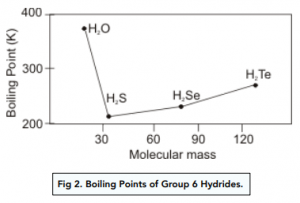
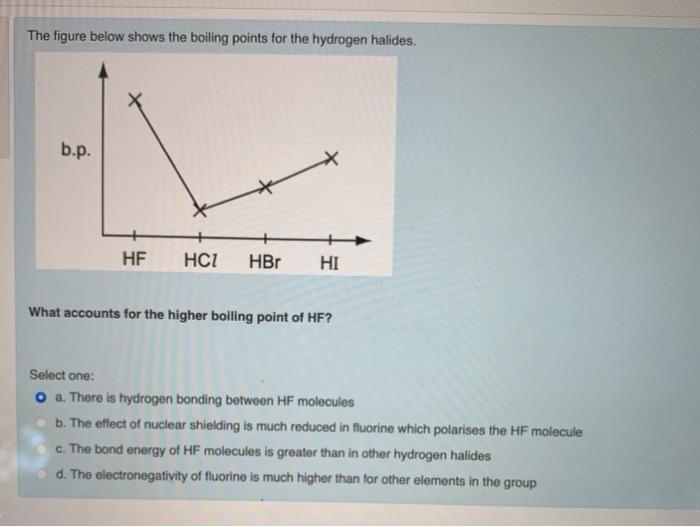
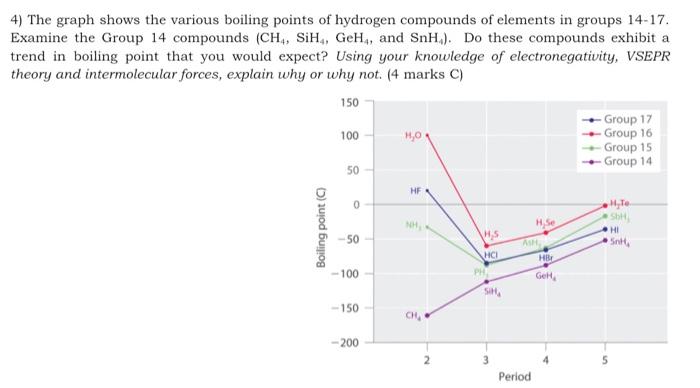
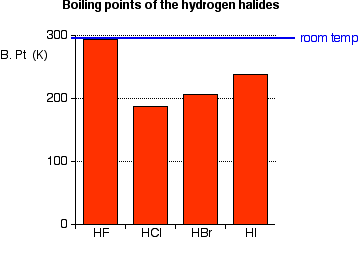
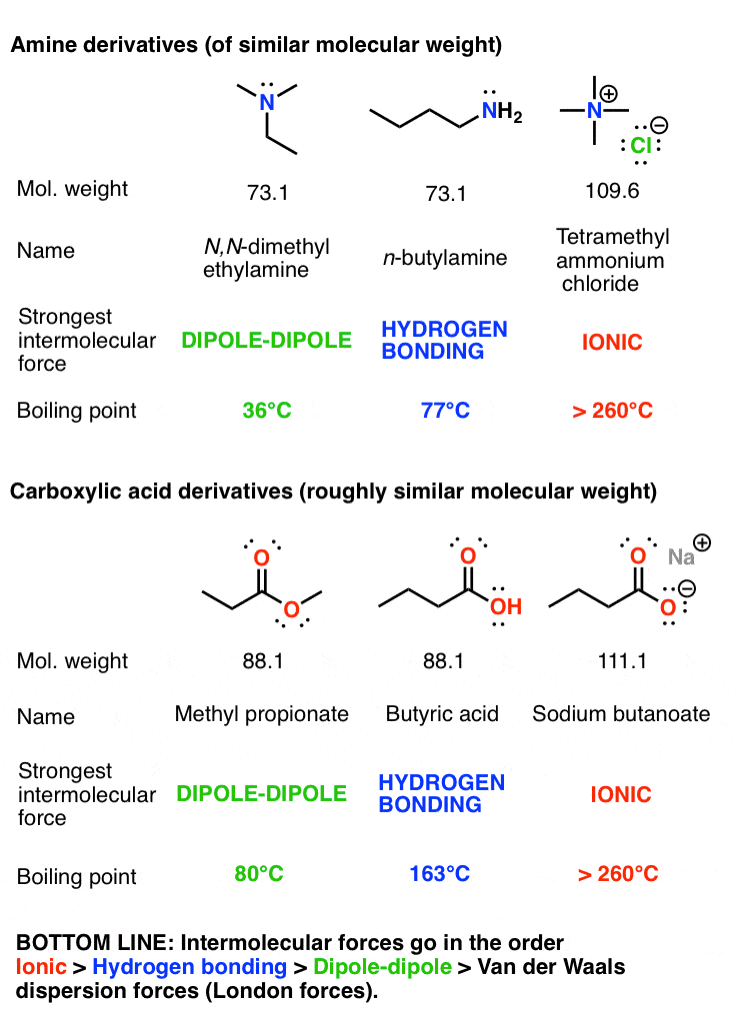
Hydrogen bonding, evidence from boiling points covalent hydride molecules water ice anomalous density hydrogen fluoride ammonia permanent dipole big difference in electronegativity propanone trichloromethane A level chemistry revision notes
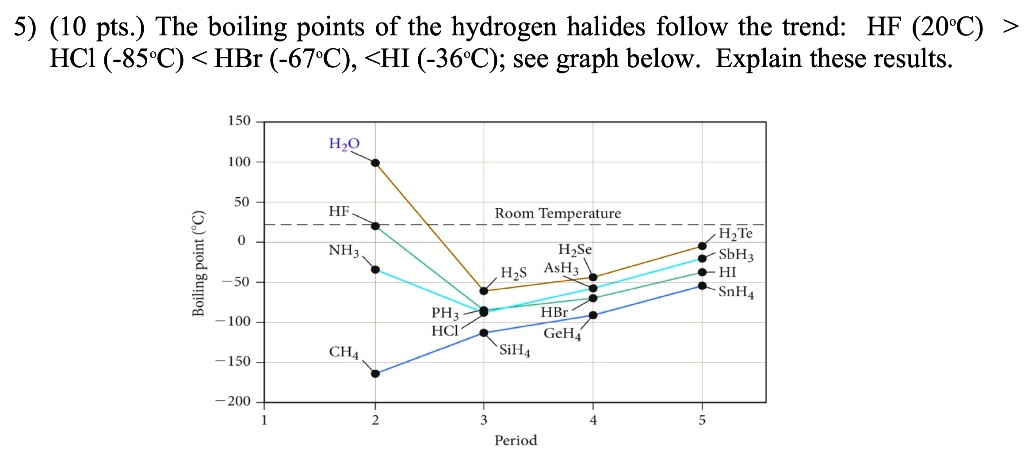
SOLVED: The boiling points of the hydrogen halides follow the trend: HF (20°C), HCl (-85°C), HBr (-67°C), HI (-36°C); see graph below. Explain these results. 150 H2O 100 HF Room Temperature 1
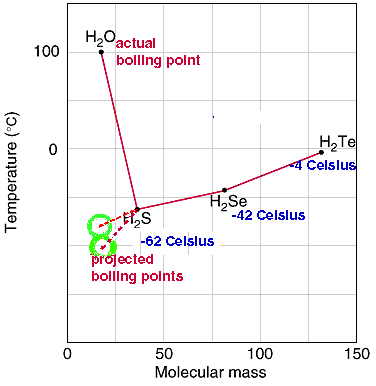
What will be the boiling point of water if hydrogen bonding between hydrogen and oxygen atoms is not present? - Quora