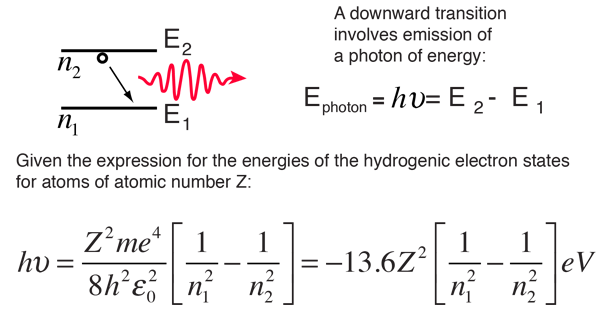
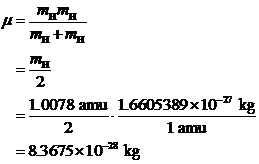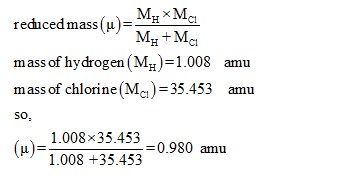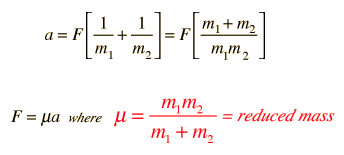SOLVED: Calculate the reduced mass for the CO molecule? Molar masses C = 12 g/mol, O = 16 g/mol. Avogadro number NA = 6.02x10^23. a. 1.81x10^-26 kg b. 2.63x10^-27 kg c. 1.14x10^-26
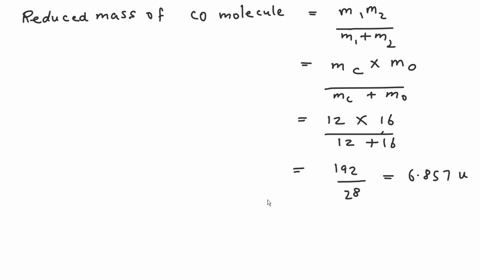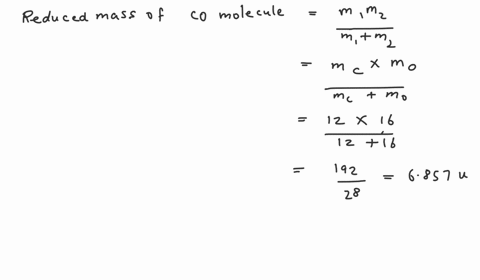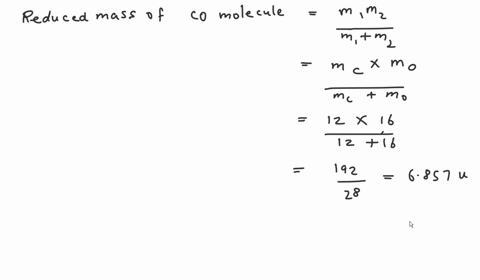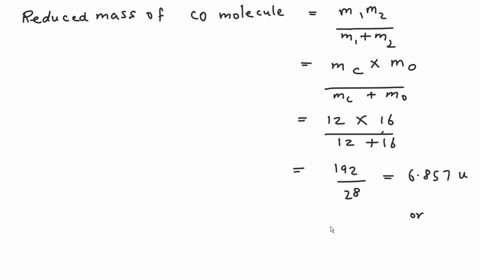
SOLVED:Calculate the reduced masses of (a) an HD molecule, (b) a ^12 C-H unit and (c) a ^13 C^16 O molecule. Exact masses: ^1 𝐇, 1.01 ;^2 H, 2.01 ;^12 C 12.00 ;^13 C, 13.00 ;^16 O, 15.99.